Rejuvenation of aged HSCs and the aged immune system in vivo
Our previous work demonstrates that aging of the immune system is driven to a large extent by aging associated changes in the function of HSCs. HSCs isolated from young or aged HSCs reconstitute distinct T- and B cell subsets that mirror young or aged immune systems. Aging of HSCs is caused by the elevated activity of the small RhoGTPase Cdc42 within HSCs. Inhibition of Cdc42 activity in aged HSCs ex vivo with a small pharmacologic inhibitor termed CASIN resulted in a sustainable rejuvenation of the function of aged HSCs. Importantly, aged HSCs treated with CASIN in vitro generated a youthful immune system capable of mounting a significantly higher immune response to vaccination compared to an immune system originated form untreated old HSCs. Thus, these experiments provide a rationale for testing CASIN in vivo treatment of aged animals to restore immune functions.
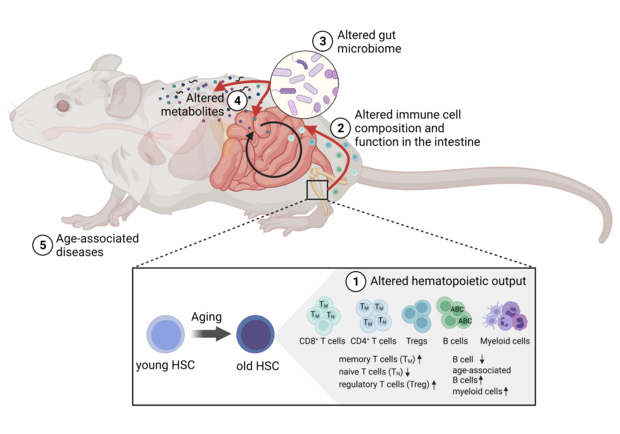
Revealing the causal relationship between age-associated immune remodeling and age-related changes in the gut microbiom
Our gut microbiota has been shown to have broad effects on our health and well-being, while changes in gut microbiota composition with age age have been linked to multiple types of diseases. Therefore, it is important to understand the causes leading to changes in gut microbiome composition with age. While it is known the gut microbiota interact with the host's immune system, causality betwwen age-related changes in immune cell populations and microbial dysbiosis has not been shown. Thus, in this project we inbestigate the novel hypothesis that age-associated changes in the immune system cause changes in the composition of the microbiome upon aging. To test this, we established a mouse model in which the age of the immune system is the prime variable in otherwise identical young animals.
Revealing the impact of HSC aging and rejuvenation on TCR Diversity of naïve T cells
Aging negatively impacts on the T cell receptor repertoire. Since the strength of an immune response correlates directly with the proportion of different antigen-specific TCRs present in the naïve T cell pool, it is thus thought that the reduced TCR clonality contributes to the decline in immune function with age. In previous work, we could show that aging of the immune system is driven to a large extent by aging of HSCs: HSCs isolated from young or old donor mice regenerated distinct immune systems resembling the ones of young or aged mice phenotypically and functionally. Importantly, HSCs, which were rejuvenated in vitro by pharmacological means, regenerated a youthful immune system with a function similar to the one found in young animals. Based on this, we will determine the clonality of T cells via sequencing approaches as a measure of determination of clonality driven by HSCs.

